 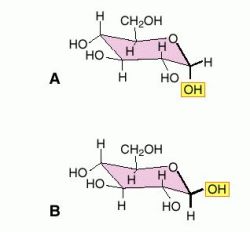
The centre of anomers in R1R2C(OH)OR, (hemiacetals R1 & R2= Hydrogen or some other organic atoms) is the anomeric carbon of position C-1. They both are relatively stereo descriptors. Types of Anomers: There are 2 types of anomers depending on their configurational arrangements in the ring alpha and beta anomers like R or S configuration in enantiomers. 
Dipolar Repulsion- This repulsion in the ring between other and anomeric groups leads to anomer destabilisation.In pyranoses (saccharide of 6-membered ring with 1 Oxygen atom and 5 carbon atoms) or other 6-membered ring shows prominent effect. 1,3-diaxial interactions- The anomer usually gets destabilised by this interaction in the axial ring.Hydrogen bonds- The hydrogen bond in the ring between other and anomeric groups leads to anomer stabilisation.This anomeric effect in polar solvents (like water) stopped. Anomeric Effect- Electron withdrawing group (especially nitrogen or oxygen atoms) in the axial ring helps stabilise the anomer.The parameters which affects stability are They possess different structures so their stabilising as well as destabilising properties also differ. Stability and Physical Properties of Anomers: All anomers differ in their stability and physical properties. The phenomenon of converting the one form of anomer into another form is known as anomerisation. An anomeric carbon is the carbon which consists of a ketone group at C-2 position or an aldehyde group at C-1 position in the open chain of the sugar moiety therefore anomers existence needs to have cyclic sugar moiety as well as it should be achiral. these are cyclic glycosides or monosaccharides which consist of different configurations at C-2 position and at C-1 position if they bear ketone and aldehyde groups in the open chain of the sugar moiety respectively. A number of other biological agents contain this motif as well.Definition: The anomers are the epimers i.e. For example, nucleosides sub-units found in DNA and RNA are all substituted at this position. It isn't an accident that in many sugar-containing biomolecules, substituents are found at the anomeric center. This position is especially attractive for nucleophiles. Furthermore, the positive charge on the oxygen brings to mind an activated carbonyl. The C=O+ unit that forms resembles a carbonyl. This mode of reaction should be familiar. Second, the anomeric center is a site of enhanced reactivity in the sugar, in terms of substitution of the carbonyl.Īnomeric reactivity involves pi donation from one oxygen to push off the other oxygen. The sugar, which is already chiral, can become either of two diastereomers when it cyclizes. This new center can form with either of two configurations. First, as you have already seen, the anomeric center is a chiral center. The anomeric center is special for two reasons. The hemiacetal position in the sugar is called the anomeric center. A second nucleophile can then donate to the pseudo-carbonyl that results. Pi donation from an oxygen in the hemiacetal can displace the other oxygen. We have already seen that hemiacetals are unstable with respect to further substitution. When a sugar cyclizes via donation of a hydroxy lone pair to the carbonyl, it forms a " hemiacetal". 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
